 
Hint: Count the number of atoms of each element, and then multiply that number by the element's atomic weight. Use the table below to find the atomic weight of each atom (element), or refer to a Periodic Table of the Elements. Let us calculate the molecular weight of some common compounds. For example, in one mole of a chemical compound there are 6.022 x 1023 molecules. Nichrome is an alloy of nickel and chromium with small amounts of silicon, manganese and iron. 
It is, however, mainly used in making alloys such as stainless steel. Nickel resists corrosion and is used to plate other metals to protect them. For example, if we are trying calculate for ammonia (NH 3 ), then we. The atomic mass is equal to the atomic number which is listed below the element symbol. One mole of 'something' contains 6.022 x 1023 entities. Thermal Conductivity0.1805 W/ (m K) Specific Heat 14300 J/ (kg K) Heat Fusion 0.558 kJ/mol. A silvery metal that resists corrosion even at high temperatures. Molar Mass of Ammonia NH 3 Step 1: The first step for calculating molar mass is to identify all the elements in a given molecule and write their atomic masses using the periodic table. A mole is the unit that measures the amount of a substance. One thousand mers connected together would add up to a weight of 28,000 grams/mole and would have 6,000 atoms.Ī mole is the standard method in chemistry for communicating how much of a substance is present. However, if we have 6. We combine (react) many mers of ethylene together to form a polyethylene chain. Relative atomic mass : Melting point (☌) Boiling point (☌) State of aggregation (20 ☌ 1013. According to the periodic table, the atomic mass of aluminum is 26.98 amu, copper is 63.55 amu, and carbon is 12.01 amu.Since 1 amu is only 1.674 × 10 24 g, these masses would be way too small to measure on ordinary laboratory equipment. 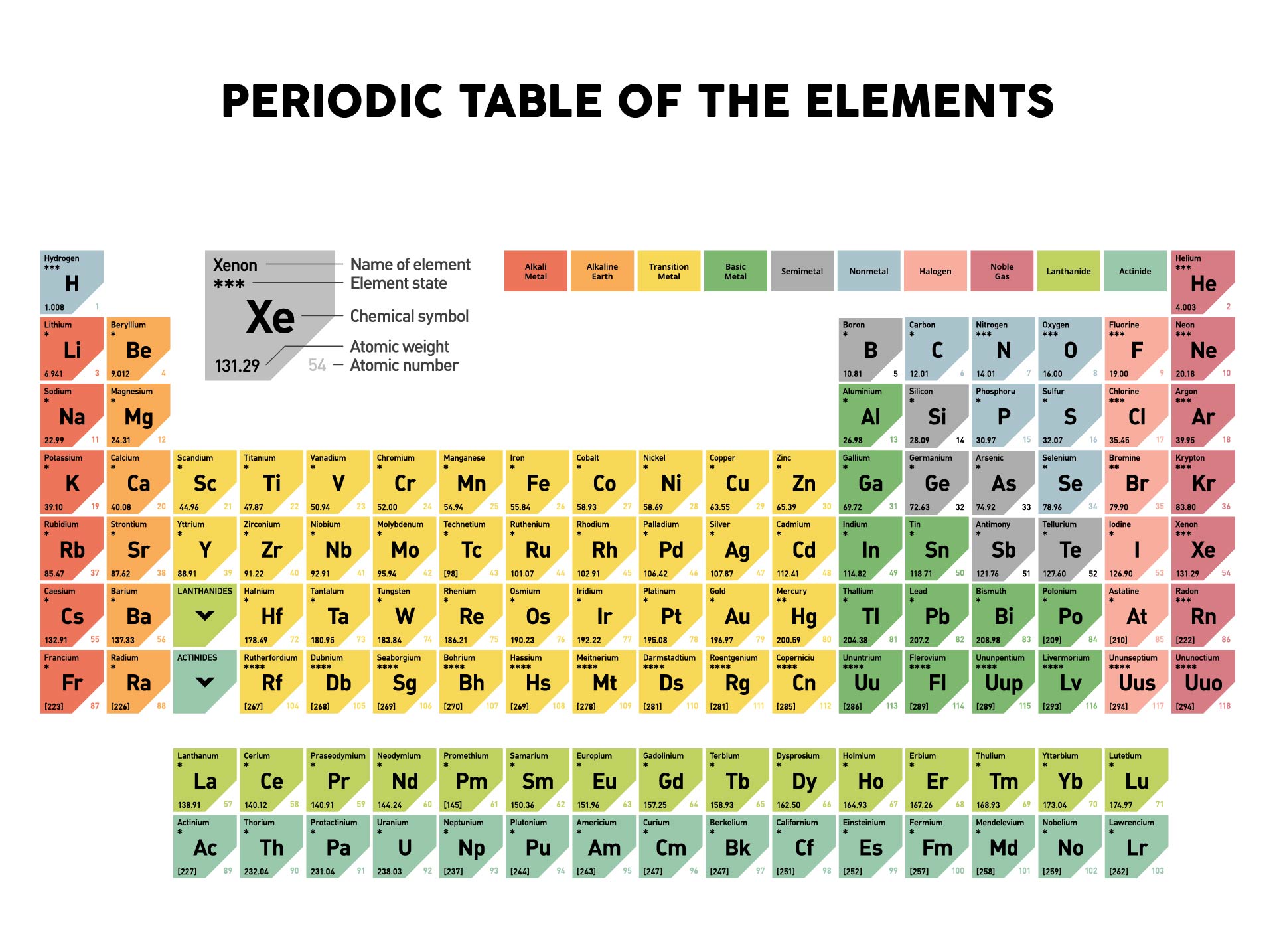
The atomic weight of carbon is 12 and that of hydrogen is 1, so one mer of ethylene has a weight of 2(12) + 4(1) = 28. It has a total of 6 atoms: 2 carbon (C) atoms and 4 hydrogen (H) atoms. The chemical formula for an ethylene monomer is -(CH2-CH2).In order to calculate the molecular weight of one water molecule, we add the contributions from each atom that is, 2(1) + 1(16) = 18 grams/mole. Using the periodic table of the elements to find atomic weights, we find that hydrogen has an atomic weight of 1, and oxygen's is 16. The chemical formula for water is H2O, which means this molecule has 3 atoms: 2 of hydrogen (H) and 1 oxygen (O) atom.No element has an atomic weight less than one. 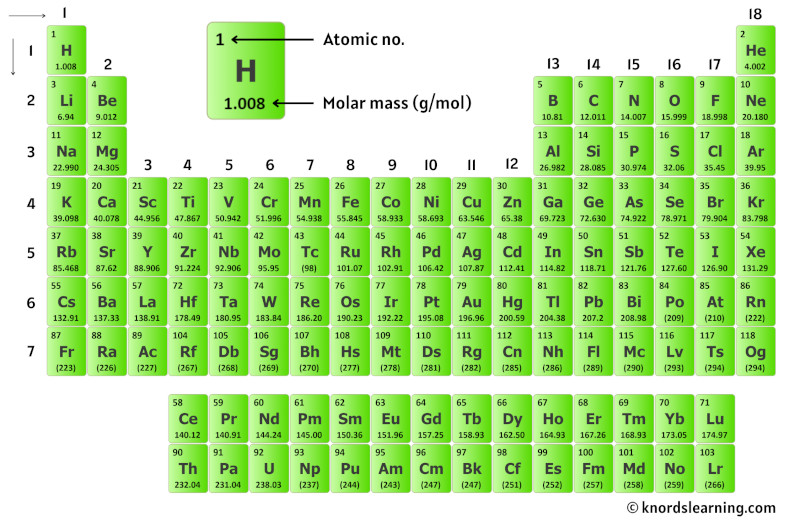
One atomic mass unit isĭefined as 1/12 the mass of an atom of carbon-12. The number in parentheses is the mass number of the most stable or best-known isotope of that element.Ītomic weights are measured in atomic mass units. These elements are unstable that is, their nuclei decompose radioactively. The atomic weight of some elements is given as a whole number enclosed in parentheses. That 75.53% of naturally occurring chlorine is chlorine-35 and 24.47% is chlorine-37. The atomic weight of chlorine (35.45) reflects the fact The atomic weight of carbon (12.01) reflects the relative abundance of The atomic weight of an element is based on the variety of naturally occurring isotopes of that element and the relative abundance of each.Ī collection of naturally occurring carbon atoms contains 98.89% carbon-12Ītoms and 1.11% carbon-13 atoms, along with a trace percentage of carbon-14Ītoms. Both the periodic table and the alphabetical list of the elements show the atomic weights of the elements. The atomic weight (or atomic mass) of an element is the average relative mass of the naturally occurring atoms of that element. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
