 
It also predicted that atoms could only emit or absorb certain wavelengths of light, which was proven correct in 1923 by Robert Andrews Millikan. He was able to apply this knowledge to every other line in the hydrogen spectrum and later extended it to all of the elements. Success and Failure of Bohr’s Atomic Model The success of Bohr’s Atomic Modelīohr’s atomic model explained why the Balmer-Rydberg formula worked so well with spectral lines. The transition between levels takes place with an extremely sharp wavefront called a wave-packet.The amount of time that the electron is in each state determines the probability that it will stay in that state or jump to another level. The electron can only absorb radiation of certain energy and only emit radiation of a higher energy level.The probability that an electron will emit or absorb a photon (i.e., to move from one state to another) is related directly to Planck’s constant and the energy difference between the two-state levels involved in the transition.A single energy state can only be occupied by one electron at a time.The electrons can only jump from one level to another in a discrete amount of time.Each energy state can only hold a specific amount of energy once an electron has gained or lost the amount of energy dictated by its current state, it needs to move onto a higher or lower level-an electron cannot stay in one state for long.The electrons are restricted to specific orbits or energy states.In order to keep the atom stable, Bohr made the following postulates: The nucleus acts as a potential barrier for an electron so that it can only transfer its orbital motion around the atom in one direction. The atomic model is a framework that determines all characteristics and interactions of the electron by its distance from the nucleus. It was based on Dalton’s work, which provided the most important step to Bohr’s model because it allowed Bohr a feasible way to analyze atoms based on the smallest possible unit of an atom. The atomic model of Niels Bohr was proposed in 1913. Bohr’s atomic model was based on an earlier model proposed by John Dalton in the early 19th century. A basic understanding of the atomic model is necessary to understand the failure of Bohr’s atomic model. Some of those models included Niels Bohr’s revised atomic model, Erwin Schrödinger’s equation, Max Planck’s constant h, and Rutherford’s experiments. Bohr’s atomic theory is still widely used today, but many different models were tried that failed to make quantum mechanics work to be a better explanation of atoms. 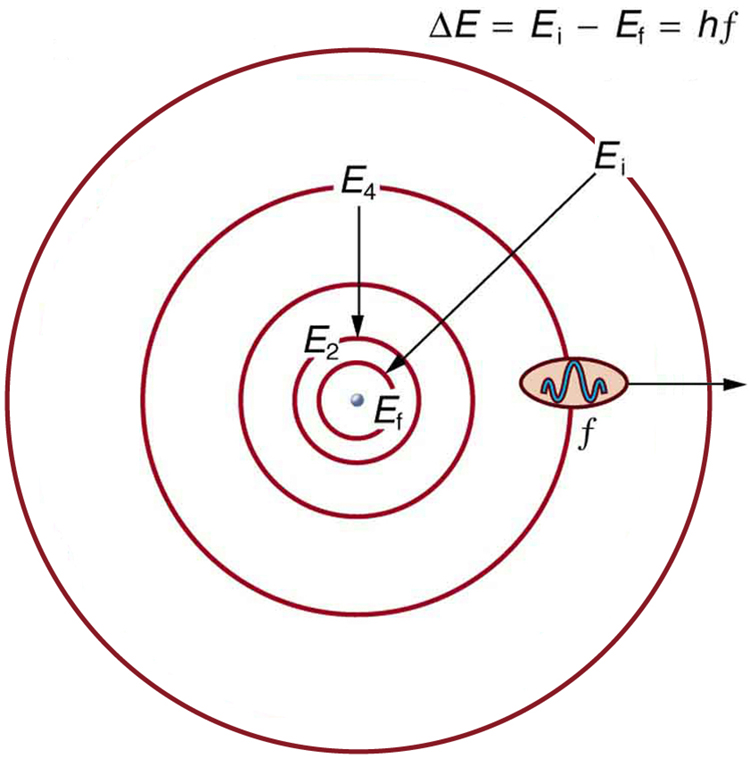 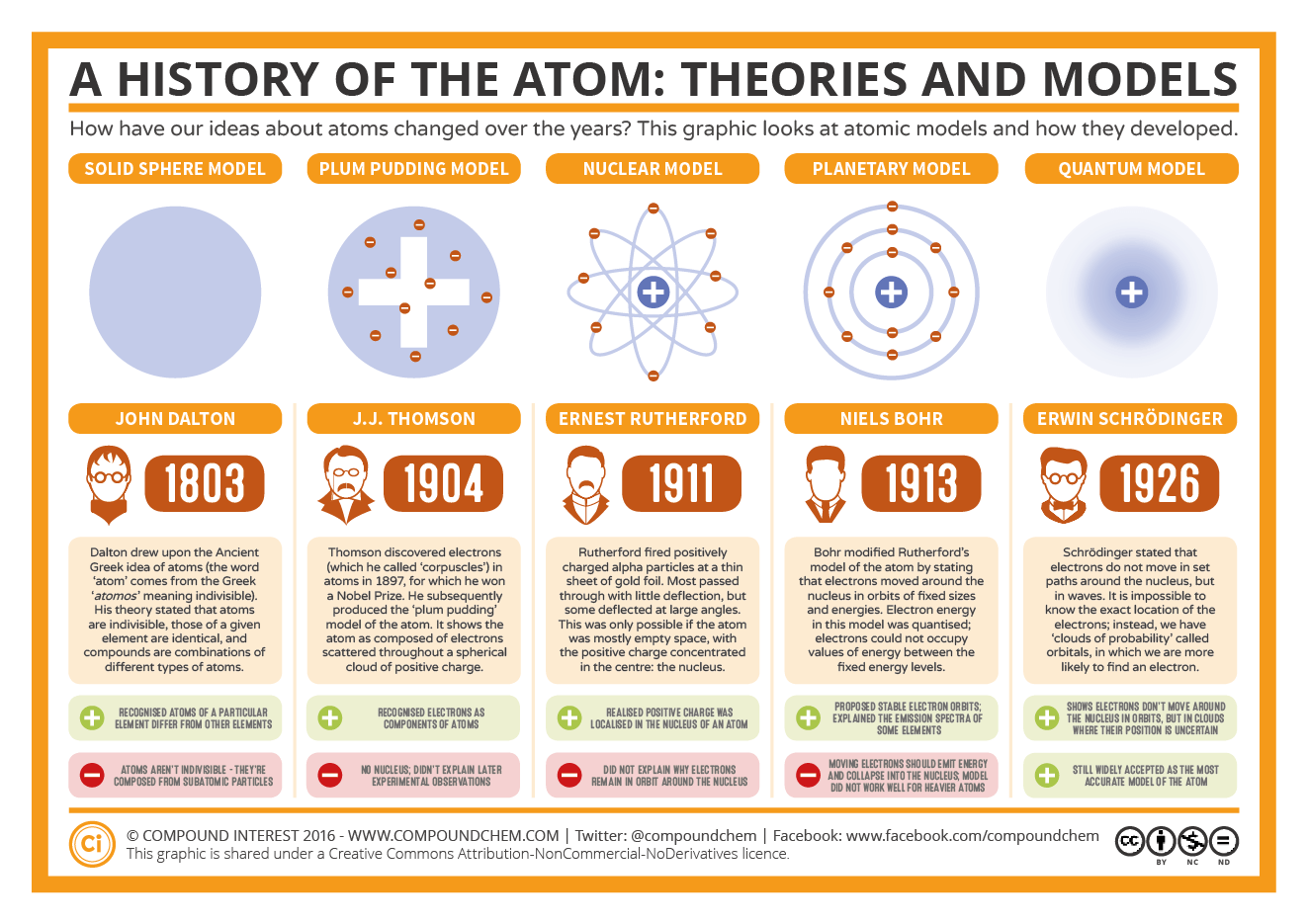
However, his model worked well as an explanation for the emissions of the hydrogen atom, but was seriously limited when applied to other atoms.Atomic theory was the best way to approach the atom until it was overthrown by quantum mechanics. Moving up the ladder increases your potential energy, while moving down the ladder decreases your energy.īohr's work had a strong influence on our modern understanding of the inner workings of the atom. As you move up or down a ladder, you can only occupy specific rungs and cannot be in the spaces in between rungs. An everyday analogy to the Bohr model is the rungs of a ladder. The electron is not allowed to occupy any of the spaces in between the orbits. The orbits that are further from the nucleus are all of successively greater energy. The ground state of the hydrogen atom, where its energy is lowest, is when the electron is in the orbit that is closest to the nucleus. When the electron is in one of these orbits, its energy is fixed. It accounts for a wide range of physical phenomena, including the existence of discrete packets of energy and matter, the uncertainty principle, and the exclusion principle.Īccording to the Bohr model, often referred to as a planetary model, the electrons encircle the nucleus of the atom in specific allowable paths called orbits. This is a theory based on the principle that matter and energy have the properties of both particles and waves. This was the basis for what later became known as quantum theory. When the energy is removed, the electrons return back to their ground state, emitting a corresponding amount of energy-a quantum of light, or photon. (Credit: Zachary Wilson Source: CK-12 Foundation License: CC BY-NC 3.0(opens in new window))īohr explained that electrons can be moved into different orbits with the addition of energy. \): Bohr's atomic model hydrogen emission spectra. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
